
 |
 |
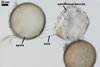
SPORES borne singly in the soil; produced laterally on the neck of a sporiferous saccule; yellowish white (3A2) to pale yellow (3A3); globose to subglobose; (90-)120(-135) µm diam.
SUBCELLULAR STRUCTURE OF SPORES consists of a spore wall and two inner germination walls.
Spore wall composed of three layers (swl1-3).
In PVLG+Melzer's reagent |
||||||
Layer 1 evanescent, hyaline, (1.0-)1.7(-2.5) µm thick, tightly adherent to layer 2, continuous with the wall of a sporiferous saccule neck, frequently completely sloughed in mature spores.
Layer 2 laminate, yellowish white (3A2) to pale yellow (3A3), (3.2-)5.5(-8.8) µm thick, ornamented with evenly distributed pits; pits circular to ellipsoid, 1.2-2.5 µm diam when seen in a plan view and 0.5-1.0 µm deep in a cross-sectional view.
Layer 3 flexible, hyaline, (0.4-)0.7(-0.8) µm thick, sometimes slightly separating from layer 2.
Layers 1-3 do not react in Melzer’s reagent.
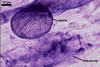
Germination wall 1 consists of two, flexible, hyaline layers (gw1l1 and 2), tightly adherent to each other in moderately crushed spores, but usually separating in vigorously crushed spores; each layer ca. 0.5 µm thick. None of these layers stains in Melzer’s reagent.
Germination wall 2 contains two layers (gw2l1 and 2).
Layer 1 flexible, (1.7-)2.2(-2.7) µm thick, covered with a granular material, frequently scattering in crushed spores.
Layer 2 plastic, 0.5-1.2 µm thick and 5.5-12.0 µm thick in spores crushed in water and PVLG, respectively, staining reddish white (8A2) in Melzer’s reagent.
GERMINATION ORB. Not found.
 |
 |
 |
 |
In PVLG+Melzer's reagent |
|||
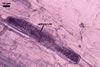
CICATRIX. The cicatrix remaining after saccule detachment is flat or slightly raised, resembling a low collar when seen in a cross-sectional view; circular to ovoid, 7.5-11.0 µm diam when observed in a plan view, with a centrally positioned pore, through which the content of a sporiferous saccule is transferred to a developing spore. At maturity, the pore is closed due to deposition of a material coming from the cytoplasm of the spore.
 |
 |
 |
In PVLG |
In PVLG+Melzer's reagent
|
|
SPORIFEROUS SACCULE hyaline; globose to subglobose; 110-140 µm diam; sometimes ovoid; 110-120 x 120-130 µm. Neck 78-85 µm long, tapering from 10.0-25.0 µm diam at the saccule to 12.5-22.5 µm diam at the spore and to 5.5-8.0 µm diam in the region of its development from a thin-walled parent hypha. Saccule wall consists of a hyaline, single, 1.7-2.5 µm thick layer, not reacting in Melzer’s reagent. Saccule collapsing at maturity because of the transfer of its content into the spore. In most mature spores, sporiferous saccule is detached.
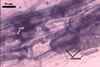
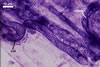
MYCORRHIZAE. The mycorrhizae of Ac. scrobiculata produced in one-species cultures with the plant host Plantago lanceolata L. consisted of arbuscules, vesicles, as well as intra- and extraradical hyphae. Arbuscules were not numerous and usually were unevenly distributed along the roots. Vesicles usually were ellipsoid, 7.5-35 x 10-52.5 µm, rarely globose, 30-38 µm, and occurred irregularly. Intraradical hyphae developed parallel to each other and to the root axis, rarely had Y-shaped branches, sometimes had short lateral branches or knobby swellings, and were 2.5-7.4 µm wide. The hyphae frequently formed coils, 15-20 x 30-40 µm. Extraradical hyphae infrequently occurred and were 2.7-2.9 µm wide. In 0.1% trypan blue, arbuscules, vesicles, coils, intra- and extraradical hyphae stained pale violet (17A3) to greyish violet (17C4), pastel violet (17B4) to light violet (17B5), violet (17A6) to greyish violet (17B5), pale violet (17A3) to greyish violet (18B5), and greyish violet (17B3-C6), respectively.
 |
 |
 |
 |
 |
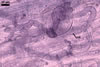 |
In roots of
P. lanceolata |
|||||
DISTRIBUTION. In Poland, spores of Ac. scrobiculata have been revealed in a trap culture containing a mixture of roots and rhizosphere soil of Corynephorus canescens (L.) P. Beauv. growing in dunes of the Baltic Sea adjacent to Swinoujscie (53o55’N, 14o14’E) in north-western Poland. The field mixture did not contain any spore of Ac. scrobiculata. Seasonal sporulation has been observed in arbuscular fungi (Gemma et al. 1989).
Acaulospora scrobiculata has originally been described from spores collected in Mexico (Trappe 1977). This fungus probably has a worldwide distribution, despite most literature reports of its finding come from the U.S.A. Literature data and the results of long-term investigations of the author of this website suggest that Ac. scrobiculata highly prefers sandy soils, especially maritime dunes. The maritime dune sites found to contain spores of Ac. scrobiculata have been those located in the U.S.A. (Friese and Koske 1991; Gemma and Koske 1989; Gemma et al. 1989; Koske 1987, 1988; Koske and Gemma 1996; Koske and Halvorson 1981; Koske and Tews 1987; Schenck and Smith 1982; Sylvia 1986; Sylvia and Will 1988; Tews and Koske 1986), Canada (Dalpé 1989), Brazil (Stürmer and Bellei 1994), Spain (including Majorca), Greece, Israel (Błaszkowski, pers. observ.), Italy (Paccioni and Puppi 1988; Puppi et al. 1986), and Australia (Koske 1975). This fungus has also been isolated from lacustrine dunes (Koske and Tews 1987). The reports of the presence of Ac. scrobiculata in non-dune soils come from the U.S.A. (Miller et al. 1985; Walker et al. 1982), Mexico (Trappe 1977), Cameroon (Musoko et al. 1994), Finland (Vestberg 1995), China (Zhang et al. 1992), Taiwan (Wu and Chen 1986), and Japan (Saito and Vergas 1991).
NOTES. Acaulospora scrobiculata distinguishes its light-coloured spores and the pitted upper surface of the laminate layer of their wall.
Eight other species of the genus Acaulospora form pitted spores. However, compared with spores of A. scrobiculata, those of the other species are either smaller (A. alpina, A. paulinae, A. taiwania, A. undulata; Błaszkowski 1988; Hu 1988; Oehl et al. 2006; Sieverding 1988) or/and larger and darker-coloured (A. cavernata, A. excavata, A. foveata, A. lacunosa, A. taiwania; Blaszkowki 1989, 2003; Hu 1988; Ingleby et al. 1994; Janos and Trappe 1982; Morton 1986, 2002). Additionally, A. taiwania produces spores in sporocarps (vs. only singly in the soil as A. scrobiculata) and the spores are ornamented with 4-5-sided pits (Hu 1988), and not pits of an arched border when seen in a plane view.
Other differences between A. scrobiculata and the other species listed above are those in the number of inner germinal walls of their spores, as well as in the phenotypic and biochemical properties of their components. Spores of A. undulata and A. taiwania have only one inner germinal wall (Hu 1988; Sieverding 1988), lacking the second germinal wall of the other Acaulospora spp. compared here. According to Oehl et al. (2006), A. undulata probably is a member of the family Archaeosporaceae. In spores of A. alpina, the upper surface of the outer layer of their second germinal wall is smooth (Oehl et al. 2006), whereas that of spores of the other species is ornamented with granular excrescences (in all but A. cavernata) or knobs (A. cavernata). Except for A. alpina, in the other species producing spores with two germinal walls, the innermost layer of the second germinal wall stains intensively in Melzer's reagent.
When devoid of a sporiferous saccule, spores of A. scrobiculata are almost indistinguishable from those of Kuklospora kentinensis (Błaszkowski, pers. observ.; Morton 2002; Sieverding and Oehl 2006; Wu et al. 1995). The ornamentation of the upper surface of the laminate spore wall layer is similar in both species. However, spores of K. kentinensis are slightly darker-coloured [pale yellow (4A3) to deep yellow (4A8) vs. yellowish white (3A2) to pale yellow (3A3) in A. scrobiculata] and may be markedly larger [85-140 x 95-210 µm vs. (90-)120(-135) µm].
The morphological properties of the A. scrobiculata mycorrhizae described here generally agree with those presented in the only earlier description of mycorrhizae of this fungus (Morton 2002). The short lateral branches and knobby swellings formed by hyphae of A. scrobiculata inside roots of P. lanceolata were not observed by Morton (2002) in roots of Zea mays. Colonization pattern of an arbuscular fungal species may be influenced by host plant (Jacquelinet-Jeanmougin and Gianinazzi-Pearson 1983; Hetrick et al. 1985).
REFERENCES
Błaszkowski J. 1988. Three new vesicular-arbuscular mycorrhizal fungi (Endogonaceae) from Poland. Bull. Pol. Ac. Sci. Biol. Sci. 36, 10-12.
Błaszkowski J. 1989. Acaulospora cavernata (Endogonaceae) – a new species from Poland with pitted spores. Crypt. Bot. 1, 204-207.
Błaszkowski J. 1990. Polish Endogonaceae. VI. Acaulospora lacunosa. Crypt. Bot. 2, 20-24.
Błaszkowski J. 1994. Polish Glomales 10. Acaulospora dilatata and Scutellospora dipurpurascens. Mycorrhiza 4, 173-182.
Błaszkowski J. 2003. Arbuscular mycorrhizal fungi (Glomeromycota), Endogone and Complexipes species deposited in the Department of Plant Pathology, University of Agriculture in Szczecin, Poland. http://www.agro.ar.szczecin.pl/~jblaszkowski/.
Dalpé Y. 1989. Inventaire et repartition de la flore endomycorhizienne de dunes et de rivages maritimes du Québec, du Nouveau-Brunswick et de la Nouvelle-Ecosse. Naturaliste Can (Rev Ecol Syst) 116, 219-236.
Friese C. F., Koske R. E. 1991. The spatial dispersion of spores of vesicular-arbuscular mycorrhizal fungi in a sand dune: microscale patterns associated with the root architecture of American beachgrass. Mycol. Res. 95, 952-957.
Gemma J. N., Koske R. E. 1989. Field inoculation of American beachgrass (Ammophila breviligulata) with V-A mycorrhizal fungi. J. Environm. Manag. 29, 173-182.
Gemma J. N., Koske R. E., Carreiro M. 1989. Seasonal dynamics of selected species of VA mycorrhizal fungi in a sand dune. Mycol. Res. 92, 317-321.
Hetrick B. A. D., Bloom J., Feyerherm S. M. 1985. Root colonization pattern of Glomus epigaeum in nine host species. Mycologia 77, 825-828.
Hu H.-T. 1988. Study on the endomycorrhizae of China fir (Cunninghamia lanceolata Hooker) and taiwania (Taiwania cryptomerioides). Q. J. Chin. For. 21, 45-72.
Ingleby K., Walker C., Mason P. A. 1994. Acaulospora excavata sp. nov. - an endomycorrhizal fungus from Cote D'Ivoire. Mycotaxon 50, 99-105.
Jacquelinet-Jeanmougin S., Gianinazzi-Pearson V. 1983. Endomycorrhizas in the gentianaceae I. The fungi associated with Gentiana lutea L. New Phytol. 95, 663-666.
Janos D. P., Trappe J. M. 1982. Two new Acaulospora species from tropical America. Mycotaxon 15, 515-522.
Koske R. E. 1975. Endogone spores in Australian sand dunes. Can. J. Bot. 53: 668-672.
Koske R. E. 1987. Distribution of VA mycorrhizal fungi along a latitudinal temperature gradient. Mycologia 79: 55-68.
Koske R. E. 1988. Vesicular-arbuscular mycorrhizae of some Hawaiian dune plants. Pacific Sci. 42, 217-229.
Koske R. E., Gemma J. N. 1996. Arbuscular mycorrhizal fungi in Hawaiian sand dunes: Island of Kaua’i. Pacific Sci. 50, 36-45.
Koske R. E., Halvorson W. L. 1981. Ecological studies of vesicular-arbuscular mycorrhizae in a barrier sand dune. Can. J. Bot. 59, 1413-1422.
Koske R. E., Tews L. L. 1987. Vesicular-arbuscular mycorrhizal fungi of Wisconsin sandy soils. Mycologia 79, 901-905.
Miller D. D., Domoto P. A., Walker C. 1985. Mycorrhizal fungi at eighteen apple rootstock plantings in the United States. New Phytol. 100, 379-391.
Morton J. B. 1986. Three new species of Acaulospora (Endogonaceae) from high aluminum, low pH soils in West Virginia. Mycologia 78, 641-648.
Morton J. B. 2002. International Culture Collection of Arbuscular and Vesicular-Arbuscular Mycorrhizal Fungi. West Virginia University. http://www.invam.caf.wvu.edu/.
Musoko M., Last F. T., Mason P. A. 1994. Populations of spores of vesicular-arbuscular mycorrhizal fungi in undisturbed soils of secondary semideciduous moist tropical forest in Cameroon. Forest Ecol. Management 63, 359-377.
Oehl F., Sýkorová Z., Redecker D., Wiemken A. 2006. Acaulospora alpina, a new arbuscular mycorrhizal fungal species characteristic for high mountainous and alpine regions of the Swiss Alps. Mycologia 98, 286-294.
Pacioni G., Puppi G. 1988. Ecologia delgi endofiti vesicicola-arbuscolari in Italia centrale. Micol. Veg. Mediter. 3, 133-142.
Puppi G., Chiapperi F., Tabacchini A., Sanvito A., Carpigo F. 1986. Endogonaceae del litorale Tirrenico. Micol. Ital. 15, 7-14.
Saito M., Vargas R. 1991. Vesicular-arbuscular mycorrhizal fungi in some humus-rich Ando soils of Japan. Soil Microorg. 38, 3-15.
Schenck N. C., Smith G. S. 1982. Additional new and unreported species of mycorrhizal fungi (Endogonaceae) from Florida. Mycologia 74,: 77-92.
Sieverding E. 1988. Two new species of vesicular arbuscular mycorrhizal fungi in the Endogonaceae from tropical high lands of Africa. Angew. Bot. 62, 373-380.
Sieverding E., Oehl F. 2006. Revision of Entrophospora and description of Kuklospora and Intraspora, two new genera in the arbuscular mycorrhizal Glomeromycetes. J. Appl. Bot. Food Qual. 80, 69-81.
Stürmer S. L., Bellei M. M. 1994. Composition and seasonal variation of spore populations of arbuscular mycorrhizal fungi in dune soils on the island of Santa Catarina, Brazil. Can. J. Bot. 72, 359-363.
Sylvia D. M. 1986. Spatial and temporal distribution of vesicular-arbuscular mycorrhizal fungi associated with Uniola paniculata in Florida foredunes. Mycologia 78, 728-734.
Sylvia D. M., Will M. E. 1988. Establishment of vesicular-arbuscular mycorrhizal fungi and other microorganisms on a beach replenishment site in Florida. Appl. Environm. Microbiol. 54, 348-352.
Tews L. A., Koske R. E. 1986. Toward a sampling stategy for vesicular-arbuscular mycorrhizas. Trans. Br. Mycol. Soc. 87, 353-358.
Trappe J. W. 1977. Three new Endogonaceae: Glomus constrictus, Sclerocystis clavispora, and Acaulospora scrobiculata. Mycotaxon 6, 359-366.
Vestberg M. 1995. Occurrence of some Glomales in Finland. Mycorrhiza 5, 329-336.
Walker C., Mize C. W., McNabb H. S. 1982. Populations of endogonaceous fungi at two localities in central Iowa. Can. J. Bot. 60, 2518-2529.
Wu C.-G., Chen Z. C. 1986. The Endogonaceae of Taiwan. I. A preliminary investigation on Endogonaceae of babmbo vegetation at Chi-Tou areas, Central Taiwan. Taiwania 31, 65-88.
Wu C.-G., Liu Y.-S., Hwuang Y.-L., Wang Y.-P., Chao C.-C. 1995. Glomales of Taiwan: V. Glomus chimonobambusae and Entrophospora kentinensis, spp. nov. Mycotaxon 53, 283-294.
Zhang M-Q., Wang Y-S., Huang L. 1992. Eight species of VA mycorrhizal fungi from northern China. Acta Mycol. Sin. 11, 258-267.